Global Leader in Plant Cell Fermentation (PCF®)
Phyton Biotech develops and applies sustainable and “green” chemistry manufacturing solutions for phytochemicals using PCF®, serving the Pharmaceutical, Traditional Chinese Medicine (TCM), Cosmetic, Agricultural and Food Ingredient industries.
Utilizing our revolutionary PCF® platform, Phyton offers a time, risk, and cost-balanced path to commercially viable production processes. With certified GMP facilities in Germany and Canada, Phyton combines highly developed technology and quality to meet our global customers’ needs. We have a successful track record of developing and implementing innovative contract development solutions for clients around the world.
What is Plant Cell Fermentation (PCF®)?
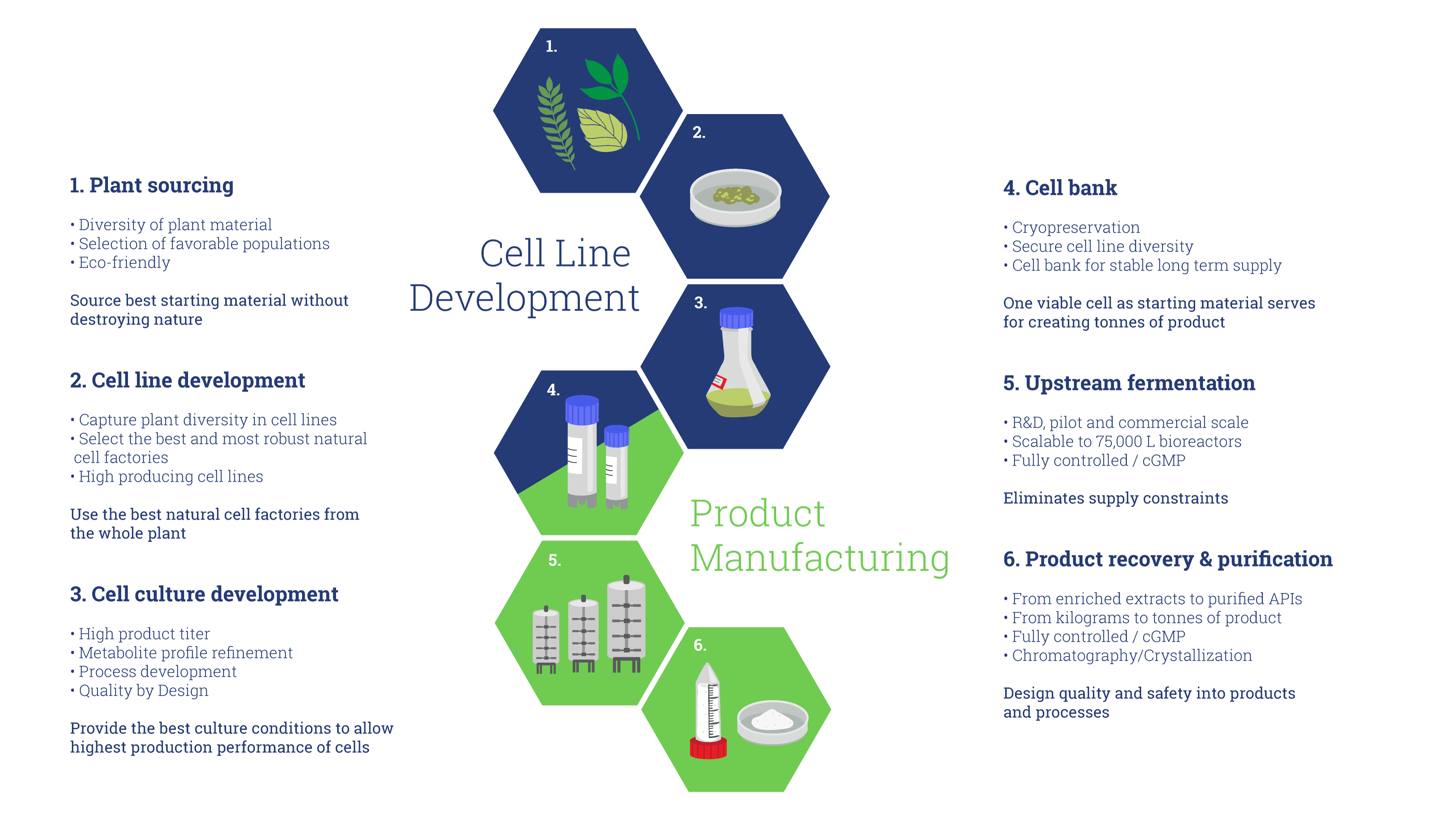
Plant Cell Fermentation (PCF®) Technology promotes the natural processes of plant cell growth and biosynthetic pathway expression ex planta in a controlled and fully defined fermentation environment:
- Non-GMO viable plant cell suspensions are used
- Starting material comes from cryopreserved cell bank
- Medium is fully defined, protein-free and does not contain animal-derived components
- Process is operated in aerated stirred tanks with proven scale-up technology
- Fermentation product can be simply extracted or further processed to concentrated active ingredient, such as for production of paclitaxel
- Fully controlled or even GMP compliant environment
Applications of Plant Cell Fermentation (PCF®)
- Supply solutions for plant-based molecules in multiple industries
- High-quality Active Pharmaceutical Ingredients (APIs) and precursors thereof
- Innovative Active Cosmetic Principles
- High-value ingredients for the flavour & fragrance industries and other food and health applications
- Safe, reliable and cost-effective way to produce high-value recombinant therapeutic proteins
Producing Phytochemicals with Plant Cell Fermentation

Why Plant Cell Fermentation (PCF®)?
PCF® provides many advantages over traditional methods of developing and producing phytochemicals.

